 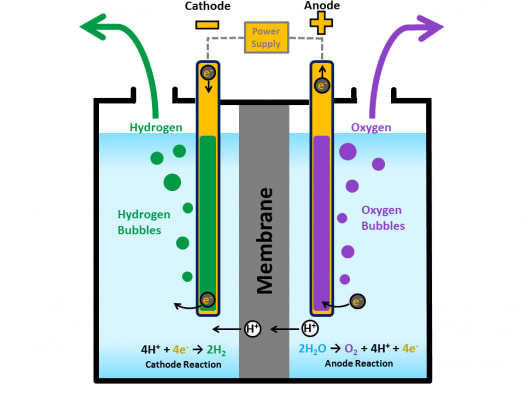
To illustrate the essential concepts of electrolysis, a few specific processes will be considered. Perhaps less familiar is the use of electrolysis in the refinement of metallic ores, the manufacture of commodity chemicals, and the electroplating of metallic coatings on various products (e.g., jewelry, utensils, auto parts). A familiar example of electrolysis is recharging a battery, which involves use of an external power source to drive the spontaneous (discharge) cell reaction in the reverse direction, restoring to some extent the composition of the half-cells and the voltage of the battery. This final section of the chapter will address an alternative scenario in which an external circuit does work on a redox system by imposing a voltage sufficient to drive an otherwise nonspontaneous reaction, a process known as electrolysis. In these cells, electrical work is done by a redox system on its surroundings as electrons produced by the redox reaction are transferred through an external circuit. Electrolysers are a critical technology for the production of low-emission hydrogen from renewable or nuclear electricity. Perform stoichiometric calculations for electrolytic processesĮlectrochemical cells in which spontaneous redox reactions take place ( galvanic cells) have been the topic of discussion so far in this chapter. The Water Electrolyzers and Fuel Cells Supply Chain Deep Dive Assessment identifies key considerations for the development of water electrolyzer and fuel cell supply chains and materials, focusing on polymer electrolyte and solid oxide technologies, to meet future demand for hydrogen produced by electrolysis and achieve U.S.  
The terminal cell voltage of an alkaline water electrolyzer is 1.82.4 V at the typical operational current density of 0.2 to 0.4 A cm 2. An electrolyzer having a partition plate produced by forming thin plates. Compare the operation of electrolytic cells with that of galvanic cells The alkaline water electrolyzer typically operates at 6080 C with a corresponding thermodynamic voltage for water splitting of 1.201.18 V. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
